Cytoprotective and immunostimulatory properties of Stachys sieboldii and Stevia rebaudiana under metabolic stress in rats
DOI:
https://doi.org/10.31489/2026feb1/32-43Keywords:
immunity, bone marrow, spleen, high-fat diet, HFHS, Stevia rebaudiana, Stachys sieboldiiAbstract
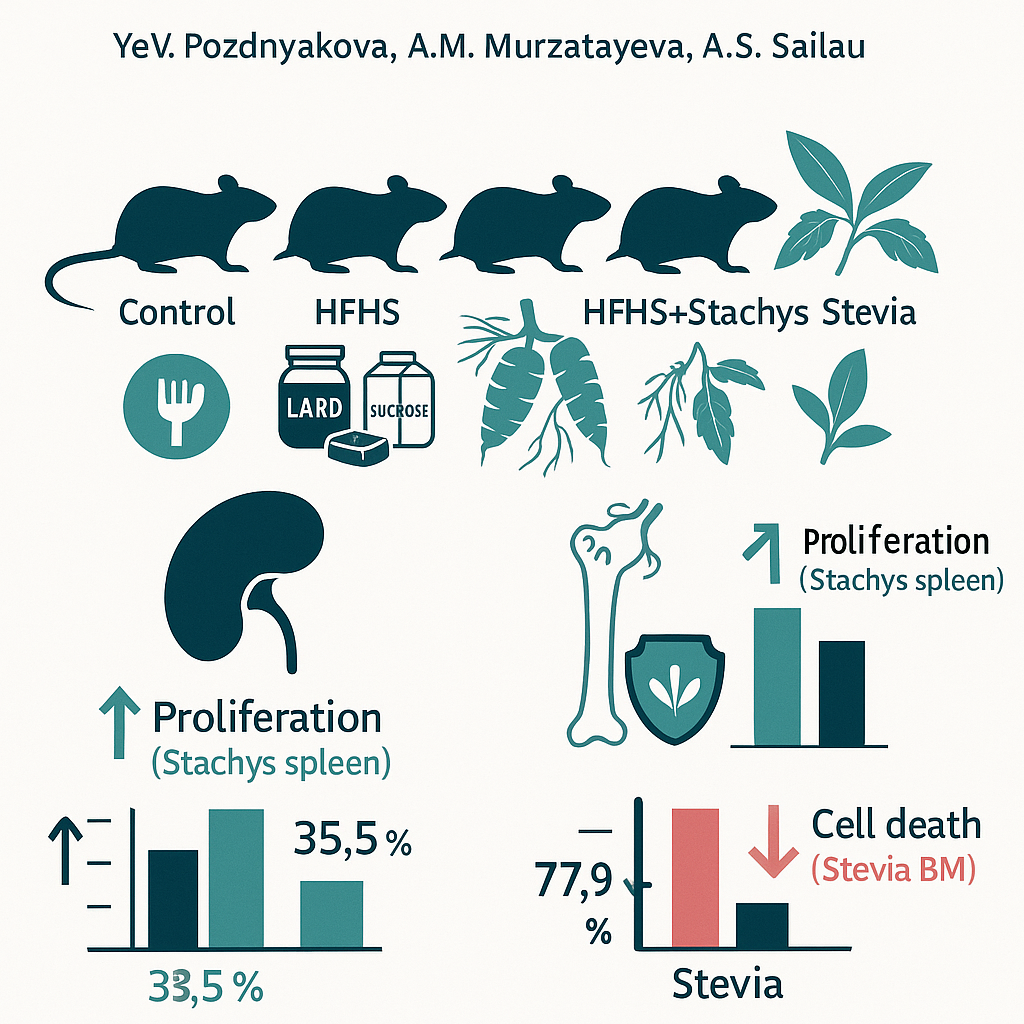
This study evaluated the protective effects of Stachys sieboldii and Stevia rebaudiana extracts on bone marrow and spleen cells in rats subjected to a high-fat, high-sucrose (HFHS) diet. Prepubertal Wistar rats were randomly assigned to four groups (control, HFHS, HFHS+Stachys, HFHS+Stevia) and maintained for 30 days. Spleen and bone-marrow cellularity and cell viability were quantified. The HFHS diet increased the proportion of non-viable cells and reduced nucleated-cell counts in both organs. Stachys primarily enhanced splenic immune-cell proliferation (higher nucleated-cell concentration), whereas Stevia produced a stronger cytoprotective response, reducing the fraction of dead cells in bone marrow. Taken together, these findings suggest complementary actions: Stachys acts as a pro-proliferative modulator of splenic immune cells, while Stevia protects hematopoietic cells from HFHS-induced damage. These results highlight the potential of these plant extracts as dietary components for supporting immune and hematopoietic function and provide a basis for their further investigation in preventive and immunomodulatory applications.
References
Goh E.V., Sobratee-Fajurally N., Allegretti A., Sardeshpande M., Mustafa M., Azam-Ali S., Omari R., Schott J., Chimonyo V., Weible D., Mutalemwa G., Mabhaudhi T., Massawe F. Transforming Food Environments: A Global lens on challenges and opportunities for achieving healthy and sustainable diets for all // Front. Sustain. Food Syst. – 2024. – Vol. 8. https://doi.org/10.3389/fsufs.2024.1366878 DOI: https://doi.org/10.3389/fsufs.2024.1366878
Clemente-Suárez V.J., Beltrán-Velasco A.I., Redondo-Flórez L., Martín-Rodríguez A., Tornero-Aguilera J.F. Global impacts of western diet and its effects on metabolism and health: a narrative review // Nutrients. – 2023. – Vol. 15, No. 12. – P. 2749. https://doi.org/10.3390/nu15122749 DOI: https://doi.org/10.3390/nu15122749
Wolfson J.A., Willits-Smith A.M., Leung C.W., Heller M.C., Rose D. Cooking at home, fast food, meat consumption, and dietary carbon footprint among US Adults // Int. J. Environ. Res. Public Health. – 2022. – Vol. 19, No. 2. – P. 853. https://doi.org/10.3390/ijerph19020853 DOI: https://doi.org/10.3390/ijerph19020853
Seale E., de Groh M., Greene-Finestone L. Fast food consumption in adults living in Canada: alternative measurement methods, consumption choices, and correlates // Appl. Physiol. Nutr. Metab. – 2023. – Vol. 48, No. 2. – P. 163–171. https://doi.org/10.1139/apnm-2022-0252 DOI: https://doi.org/10.1139/apnm-2022-0252
Baker P., Machado P., Santos T., Sievert K., Backholer K., Hadjikakou M., Russell C., Huse O., Bell C., Scrinis G., Worsley A., Friel S., Lawrence M. Ultra-processed foods and the nutrition transition: global, regional, and national trends, food systems transformations and political economy drivers // Obes. Rev. – 2020. – Vol. 21, No. 12. – P. e13126. https://doi.org/10.1111/obr.13126 DOI: https://doi.org/10.1111/obr.13126
Patel O., Shahulhameed S., Shivashankar R., Tayyab M., Rahman A., Prabhakaran D., Tandon N., Jaacks L.M. Association between full service and fast food restaurant density, dietary intake and overweight/obesity among adults in Delhi, India // BMC Public Health. – 2017. – Vol. 18, No. 1. – P. 36. https://doi.org/10.1186/s12889-017-4598-8 DOI: https://doi.org/10.1186/s12889-017-4598-8
Wu Y., Wang L., Zhu J., Gao L., Wang Y. Growing fast food consumption and obesity in Asia: challenges and implications // Soc. Sci. Med. – 2021. – Vol. 269. – P. 113601. https://doi.org/10.1016/j.socscimed.2020.113601 DOI: https://doi.org/10.1016/j.socscimed.2020.113601
Powell-Wiley T.M., Poirier P., Burke L.E., Després J.P., Gordon-Larsen P., Lavie C.J., Lear S.A., Ndumele C.E., Neeland I.J., Sanders P., St-Onge M.P. Obesity and cardiovascular disease: a scientific statement from the American Heart Association // Circulation. – 2021. – Vol. 143, No. 21. – P. e984–e1010. https://doi.org/10.1161/CIR.0000000000000973 DOI: https://doi.org/10.1161/CIR.0000000000000973
Koliaki C., Dalamaga M., Liatis S. Update on the obesity epidemic: after the sudden rise, is the upward trajectory beginning to flatten? // Curr. Obes. Rep. – 2023. – Vol. 12, No. 4. – P. 514–527. https://doi.org/10.1007/s13679-023-00527-y DOI: https://doi.org/10.1007/s13679-023-00527-y
Li Y., Zhu S., Zhang Y., Liu T., Su L., Zhang Q., Luo Y. High fat diet-induced obesity exacerbates hematopoiesis deficiency and cytopenia caused by 5-fluorouracil via peroxisome proliferator-activated receptor γ // Exp. Hematol. – 2018. – Vol. 60. – P. 30–39.e1. https://doi.org/10.1016/j.exphem.2017.12.013 DOI: https://doi.org/10.1016/j.exphem.2017.12.013
Nelson B.N., Friedman J.E. Developmental programming of the fetal immune system by maternal western-style diet: mechanisms and implications for disease pathways in the offspring // Int. J. Mol. Sci. – 2024. – Vol. 25, No. 11. – P. 5951. https://doi.org/10.3390/ijms25115951 DOI: https://doi.org/10.3390/ijms25115951
Carrillo-Martinez E.J., Flores-Hernández F.Y., Salazar-Montes A.M., Nario-Chaidez H.F., Hernández-Ortega L.D. Quercetin, a flavonoid with great pharmacological capacity // Molecules. – 2024. – Vol. 29, No. 5. – P. 1000. https://doi.org/10.3390/molecules29051000 DOI: https://doi.org/10.3390/molecules29051000
El-Kazaz S.E., Hafez M.H., Albadrani G.M., Al-Ghadi M.Q., Abdel-Daim M.M., El-Sayed Y.S. The influence of quercetin on behavior, performance and splenic immunity in broiler chickens // Vet. Anim. Sci. – 2024. – Vol. 26. – P. 100398. https://doi.org/10.1016/j.vas.2024.100398 DOI: https://doi.org/10.1016/j.vas.2024.100398
Farag M.R., Moselhy A.A.A., El-Mleeh A., Aljuaydi S.H., Ismail T.A., Di Cerbo A., Crescenzo G., Abou-Zeid S.M. Quercetin alleviates the immunotoxic impact mediated by oxidative stress and inflammation induced by doxorubicin exposure in rats // Antioxidants. – 2021. – Vol. 10, No. 12. – P. 1906. https://doi.org/10.3390/antiox10121906 DOI: https://doi.org/10.3390/antiox10121906
Khattab H.A.H., Abounasef S.K., Bakheet H.L. The Biological and Hematological Effects of Echinacea purpurea L. roots extract in the immunocompromised rats with cyclosporine // J. Microsc. Ultrastruct. – 2019. – Vol. 7, No. 2. – P. 65–71. https://doi.org/10.4103/JMAU.JMAU_62_18 DOI: https://doi.org/10.4103/JMAU.JMAU_62_18
Sullivan A.M., Laba J.G., Moore J.A., Lee T.D. Echinacea-induced macrophage activation // Immunopharmacol. Immunotoxicol. – 2008. – Vol. 30, No. 3. – P. 553–574. https://doi.org/10.1080/08923970802135534 DOI: https://doi.org/10.1080/08923970802135534
Fu A., Wang Y., Wu Y., Chen H., Zheng S., Li Y., Xu X., Li W. Echinacea purpurea extract polarizes M1 macrophages in murine bone marrow-derived macrophages through the activation of JNK // J. Cell. Biochem. – 2017. – Vol. 118, No. 9. – P. 2664–2671. https://doi.org/10.1002/jcb.25875 DOI: https://doi.org/10.1002/jcb.25875
Rimmelé P., Lofek-Czubek S., Ghaffari S. Resveratrol increases the bone marrow hematopoietic stem and progenitor cell capacity // Am. J. Hematol. – 2014. – Vol. 89, No. 12. – P. E235–E238. https://doi.org/10.1002/ajh.23837 DOI: https://doi.org/10.1002/ajh.23837
Inchingolo A.D., Inchingolo A.M., Malcangi G., Avantario P., Azzollini D., Buongiorno S., Viapiano F., Campanelli M. Effects of resveratrol, curcumin and quercetin supplementation on bone metabolism – a systematic review // Nutrients. – 2022. – Vol. 14, No. 17. – P. 3519. https://doi.org/10.3390/nu14173519 DOI: https://doi.org/10.3390/nu14173519
Gao X., Xu Y.X., Janakiraman N., Chapman R.A., Gautam S.C. Immunomodulatory activity of resveratrol: suppression of lymphocyte proliferation, development of cell-mediated cytotoxicity, and cytokine production // Biochem. Pharmacol. – 2001. – Vol. 62, No. 9. – P. 1299–1308. https://doi.org/10.1016/s0006-2952(01)00775-4 DOI: https://doi.org/10.1016/S0006-2952(01)00775-4
Tomou E.M., Barda C., Skaltsa H. Genus Stachys: A review of traditional uses, phytochemistry and bioactivity // Medicines. – 2020. – Vol. 7, No. 10. – P. 63. https://doi.org/10.3390/medicines7100063 DOI: https://doi.org/10.3390/medicines7100063
Benedec D., Oniga I., Hanganu D., Tiperciuc B., Nistor A., Vlase A.M., Vlase L., Pușcaș C., Duma M., Login C.C., Niculae M., Silaghi-Dumitrescu R. Stachys species: comparative evaluation of phenolic profile and antimicrobial and antioxidant potential // Antibiotics. – 2023. – Vol. 12, No. 11. – P. 1644. https://doi.org/10.3390/antibiotics12111644 DOI: https://doi.org/10.3390/antibiotics12111644
Peteliuk V., Rybchuk L., Bayliak M., Storey K.B., Lushchak O. Natural sweetener stevia rebaudiana: functionalities, health benefits and potential risks // EXCLI J. – 2021. – Vol. 20. – P. 1412–1430. https://doi.org/10.17179/excli2021-4211
Patel S., Navale A. The Natural Sweetener Stevia: an updated review on its phytochemistry, health benefits, and anti-diabetic study // Curr. Diabetes Rev. – 2024. – Vol. 20, No. 2. – P. e010523216398. https://doi.org/10.2174/1573399819666230501210803 DOI: https://doi.org/10.2174/1573399819666230501210803
Ajami M., Seyfi M., Abdollah Pouri Hosseini F., Naseri P., Velayati A., Mahmoudnia F., Zahedirad M., Hajifaraji M. Effects of stevia on glycemic and lipid profile of type 2 diabetic patients: a randomized controlled trial // Avicenna J. Phytomed. – 2020. – Vol. 10, No. 2. – P. 118–127. https://doi.org/32257884
Gins M.S., Gins V.K., Kononkov P.F. (2015) Stachys is a promising vegetable crop with officinal properties. biochemical and farmacological properties. Vegetable crops of Russia., 3(4), 108-112. (In Russ.) https://doi.org/10.18619/2072-9146-2015-3-4-108-112 DOI: https://doi.org/10.18619/2072-9146-2015-3-4-108-112
Hsieh M.H., Chan P., Sue Y.M., Liu J.C., Liang T.H., Huang T.Y., Tomlinson B., Chow M.S., Kao P.F., Chen Y.J. Efficacy and tolerability of oral stevioside in patients with mild essential hypertension: a two-year, randomized, placebo-controlled study // Clin. Ther. – 2003. – Vol. 25, No. 11. – P. 2797–2808. https://doi.org/10.1016/s0149-2918(03)80334-x DOI: https://doi.org/10.1016/S0149-2918(03)80334-X
Polianskaia G.G., Efremova T.N., Koltsova A.M., Musorina A.S., Sharlaimova N.S., Iakovleva T.K. A Training manual for working with cell cultures of animals and humans // Politech-Press. – 2019. – P. 114.
Othman F., El-Sawi M., Shabana S. Stevia extract safeguards rats from diabetic renal injury by mitigating markers of inflammation, oxidative stress, and apoptosis // Toxicol. Environ. Health Sci. – 2023. – Vol. 15. https://doi.org/10.1007/s13530-023-00188-3 DOI: https://doi.org/10.1007/s13530-023-00188-3
Cai T., Ye H., Jiang H., Lin C., Lou C., Wang W., Yan Z., Xue X., Pan X., Lin J. Stevioside targets the NF-κB and MAPK pathways for inhibiting inflammation and apoptosis of chondrocytes and ameliorates osteoarthritis in vivo // Int. Immunopharmacol. – 2023. – Vol. 115. – P. 109683. https://doi.org/10.1016/j.intimp.2023.109683 DOI: https://doi.org/10.1016/j.intimp.2023.109683
Gupta E., Mohammed A., Mishra N., Purwar S., Syed I., Rizvi S.I., Sundaram S. Antioxidant and anti-diabetic potential of rebaudioside A and a mixture of steviol glycosides in alloxan-induced diabetic rats // Indian J. Nat. Prod. Resour. – 2021. – Vol. 12. – P. 391–399.
Moubder E., Abass H., Al-Mahdawi M., Abd F. Immunomodulatory effects of Stevia rebaudiana leaves and commercial stevia on rats: a comparative study // Rev. Clin. Pharmacol. Pharmacokinet. – 2024. – Vol. 38. – P. 153–156. https://doi.org/10.61873/IVJV6786 DOI: https://doi.org/10.61873/IVJV6786
Kim H.H., Jeong S.H., Park M.Y., Bhosale P.B., Abusaliya A., Lee S.J., Heo J.D., Kim H.W., Seong J.K., Kim D.I., Park K.I., Kim G.S. Binding affinity screening of polyphenolic compounds in Stachys affinis extract (SAE) for their potential antioxidant and anti-inflammatory effects // Sci. Rep. – 2024. – Vol. 14, No. 1. – P. 18095. https://doi.org/10.1038/s41598-024-68880-z DOI: https://doi.org/10.1038/s41598-024-68880-z
Slimani W., Maioli M., Cruciani S., Zerizer S., Santaniello S., Kabouche Z., Coradduzza D., Chessa M., Fancello S., Migheli R., Serra P.A., D'hallewin G. Antioxidant, anti-inflammatory and anti-proliferative properties of Stachys circinata on HepG2 and MCF7 cells // Plants. – 2023. – Vol. 12, No. 12. – P. 2272. https://doi.org/10.3390/plants12122272 DOI: https://doi.org/10.3390/plants12122272
Bayat E., Rahpeima Z., Dastghaib S., Gholizadeh F., Erfani M., Asadikaram G., Mokarram P. Stevia rebaudiana extract attenuates metabolic disorders in diabetic rats via modulation of glucose transport and antioxidant signaling pathways and aquaporin-2 expression in two extrahepatic tissues // J. Food Biochem. – 2020. – Vol. 44, No. 8. – P. e13252. https://doi.org/10.1111/jfbc.13252 DOI: https://doi.org/10.1111/jfbc.13252